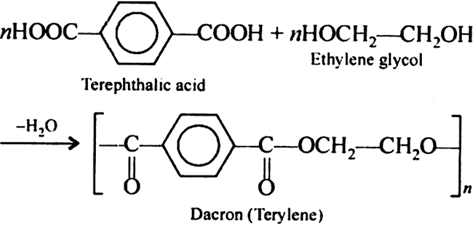
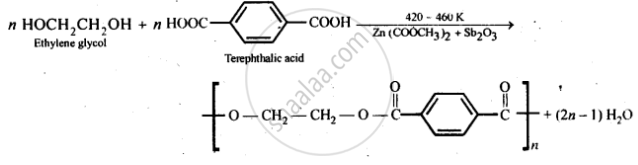
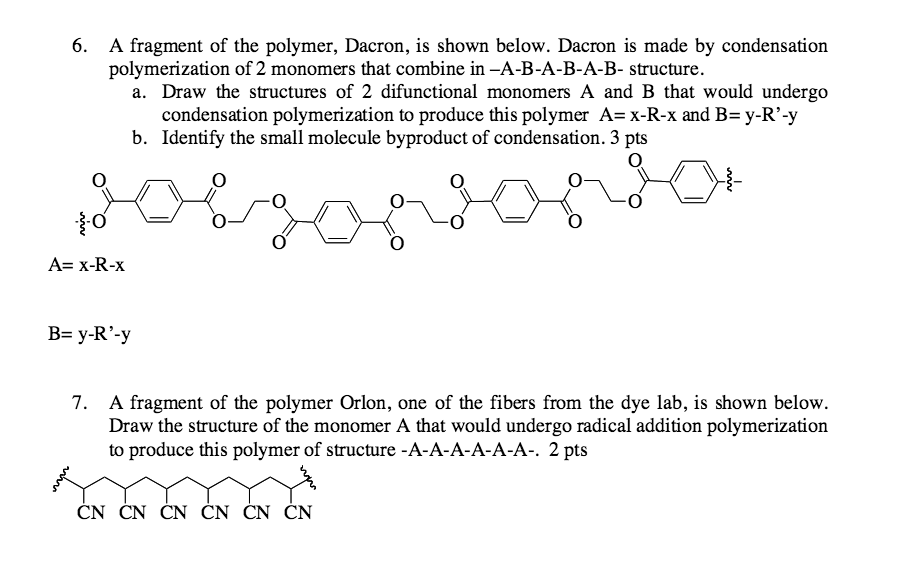
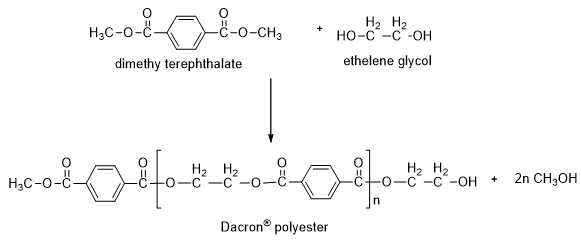
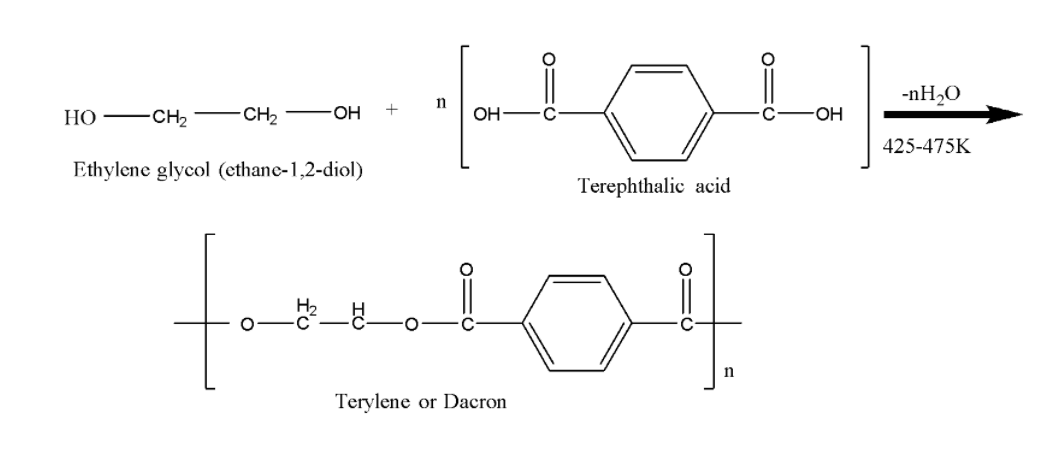
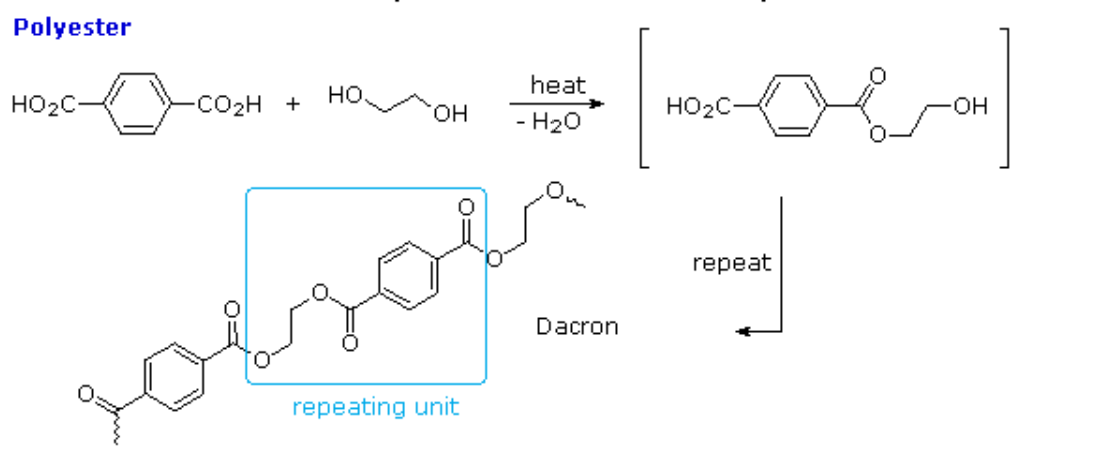
Give the monomer of dacron and process of polymerisation of dacron. - Sarthaks eConnect | Largest Online Education Community
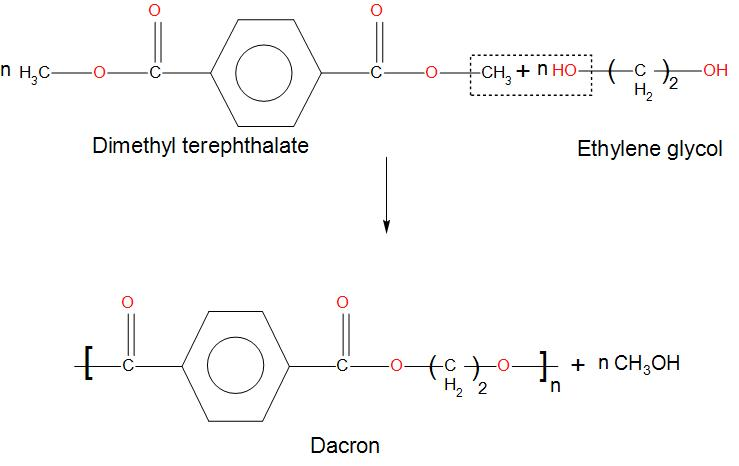
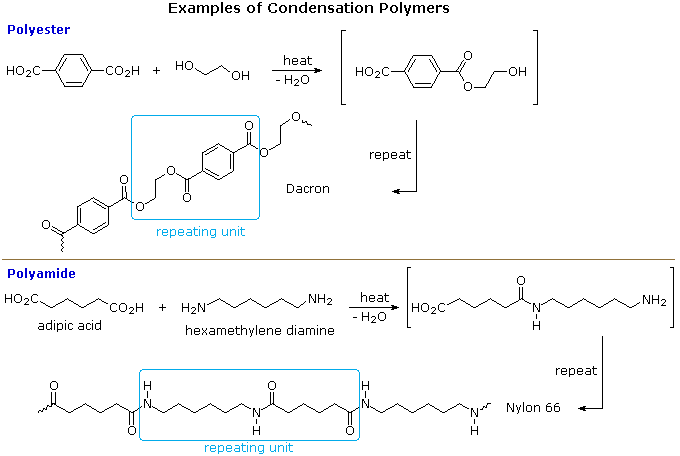
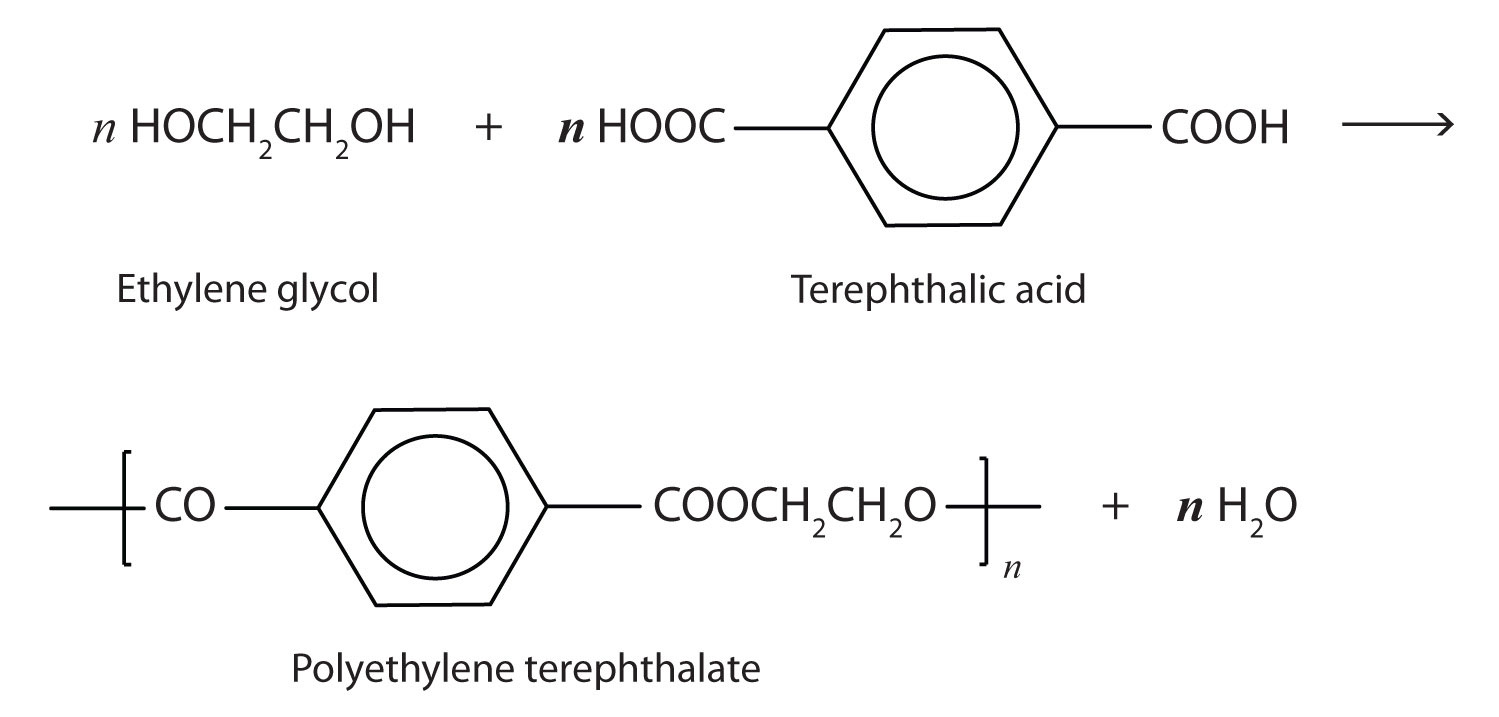
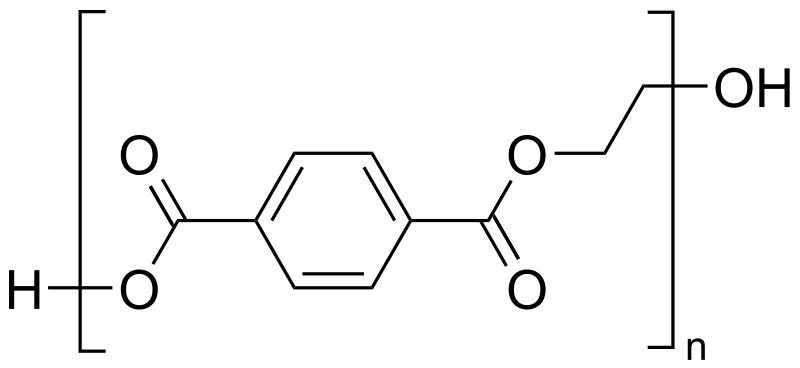
Dacron is obtained by the condensation polymerization of:(A) dimethyl terephthalate and ethylene glycol(B) terephthalic acid and ethylene glycol(C) phenol and phthalic acid(D) phenol and formaldehyde

Polymers - Condensation Polymerization || Polyesters Nylon Bakelite Melamine ||NEET JEE JAM GATE NET - YouTube

Synthetic Condensation polymers: Dacron & Nylon 6AS Group 3 Kitty Au-Yeung Giselle Ho Rachel Chui Cici Tam Angela Tsui. - ppt download

Example for polyester Dacron formed from condensation polymerization of... | Download Scientific Diagram
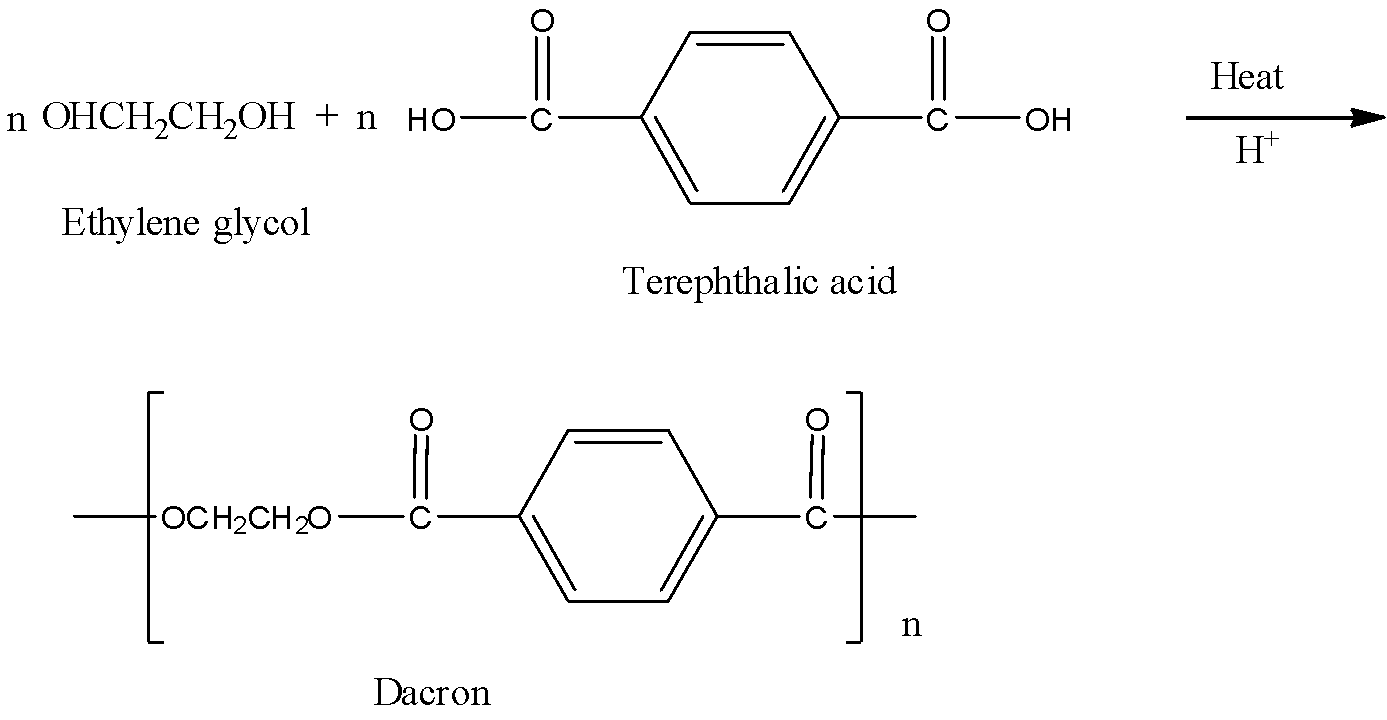
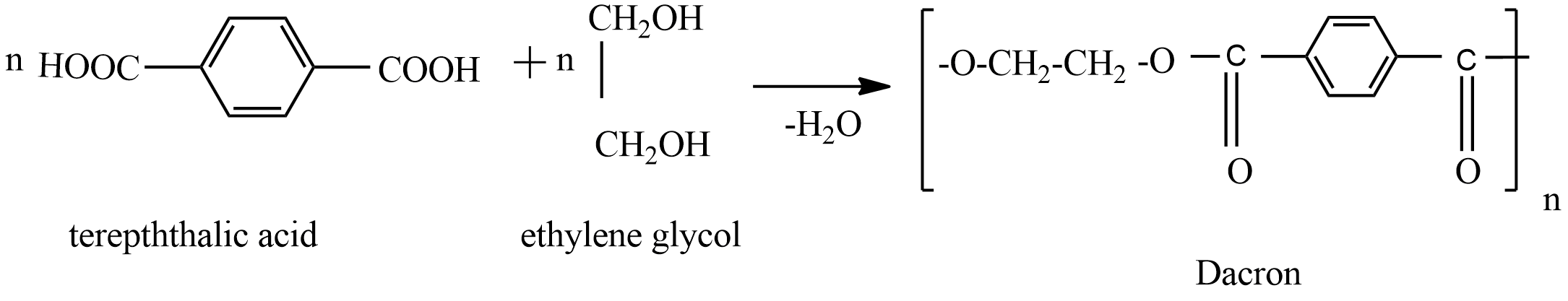
Dacron is polymer of:A) Glycol and formaldehydeB) Glycol and phenolC) Glycol and phthalic acidD) Glycol and terephthalic acid